Best CDSCO Medical Device Consulting Services in Kanpur, Uttar Pradesh
Lighten Your Load in CDSCO Medical Device Licensing & Registration
Are you searching for reliable CDSCO Medical Device Consultant in Kanpur, Uttar Pradesh? Look no further than MedDev Experts, your trusted partner for obtaining CDSCO Medical Device Licenses. With a stellar reputation and a proven track record of success, we are committed to delivering exceptional services to our esteemed clients.
WELCOME TO
Simplify the intricate process of medical device licensing and registration with the expertise of MedDev Experts. Our team of experienced professionals provides comprehensive guidance, ensuring compliance with all requirements and facilitating the smooth acquisition of necessary licenses.
- Medical Device Manufacturing License: Register your medical devices with the CDSCO and State FDA of Uttar Pradesh effortlessly.
- Medical Device Import License: Obtain the CDSCO Medical Device Import License required for importing medical devices into India.
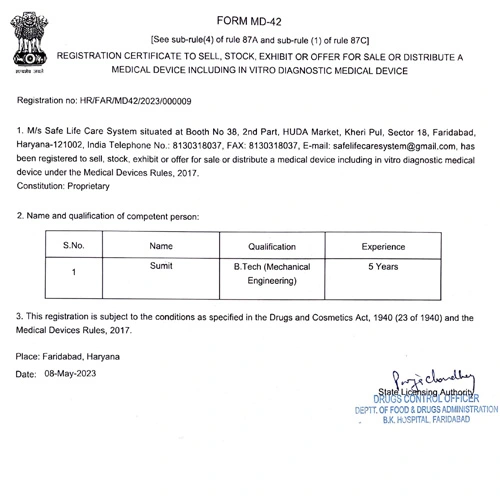
- Wholesale License for Medical Device: Secure the registration certificate to sell, stock, exhibit, or distribute medical devices.
- ISO Certification: Attain ISO 9001, ISO 13485, SA 8000, and other international quality standards with our assistance.
- GMP Compliance: Ensure your manufacturing process adheres to Good Manufacturing Practices (GMP) with our expert guidance.
- FDA Product Listing: List your medical device on the US FDA, facilitating exports to the US market.
- 510(k) Submission: Submit the Premarket Submission to the FDA for market placement in the US.
Take the first step today and discover how MedDev Experts can support you in meeting your medical device licensing and registration needs. Experience peace of mind as our dedicated team navigates the intricate process on your behalf, ensuring your success.
Successful Licensing Partnerships: Companies We’ve Empowered.


Are you ready to start your medical device licensing journey? Our team of experts can help you navigate the complex regulatory process and get your product to market quickly and efficiently.




